Lighting technology
Most sources of light are heatemitters. If you heat an object enough eventually it will start glowing red. If you heat it further it will glow yellow, then white and finally blue. It emits a continuous spectrum of electromagnetic waves, with a peak effect that is dependent on the temperature. The spectral qualities of the light that an ideal black mass emits at a specified temperature is defined as color temperature. Noon sunlight is about 5000° K (K=Kelvin, which is the temperature in celcius + 273°), since the sun also is a heatemitter. Lower color temperatures, that create red to yellow light are produced by flames normal incandescent lamps and halogen bulbs. Higher color temperatures, that create blue light can be seen in the shade of a noontime sun (lighting from the blue sky) and shortly after sunset, especially in a snow covered area. In the development of lamps it is important to achieve a maximum temperature of the element. This, in addition to raising the color temperature, also increases the amount of light and thereby the efficiency. Unfortunately raising the temperature also means that the element becomes depleted by materials boiling off it that now find a new permanent location on the first colder surface that they find (the inside of the glass bulb). This process continues until enough material is lost from the element for it to break.
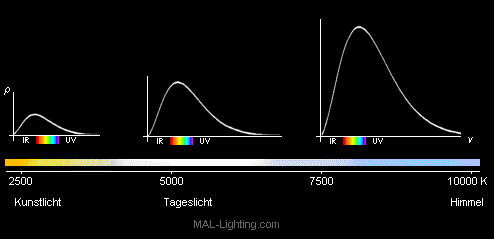 |
Since the element is already made out of tungstin, no other usable materials with a higher melting point and lower boiling pressure are available. Lamps that are overdriven (voltage too high) deliver higher color temperature and efficiency at the cost of shortened lifespans. To create lamps with long lifespans and high efficiency, you are either forced to use a chemical or physical trick, or use the semiconductor technology.
Light quantity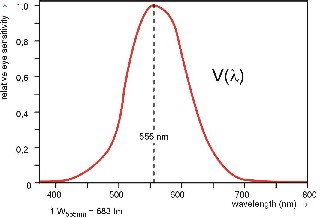
The total emissions in all directions of a lighting element.. These emissions are weighted according to the sensitivity of the human eye for various color temperatures (as per DIN 5031 Part 3). The light quantity is measured in Lumens (lm). For example: light at a wavelength of 555 Nanometer (555 Billionths of a Meter) and an emitted energy of 1 Watt produces a light quantity of 683 Lumens. Visable light has a wavelength between 380 nm (violet, wavelengths between 100 and 380 nm are ultraviolet) and 780 nm (red, wavelengths between 780 nm and 1 mm are infrared). 555 nm is about the middle of the visable range and we see a yellow-greenish light.